Industry News
Curation by industry experts
Hot Articles
Popular Tags
How pharmaceutical cold chain audits miss packaging weak points
Pharmaceutical Cold Chain audits often verify temperatures, documents, and routes, yet overlook packaging weak points that quietly undermine product safety. For researchers, operators, QA teams, and enterprise decision-makers, this gap affects not only Cold-Chain Infrastructure performance but also broader Food Storage Solutions and Cryogenic Storage strategies. Understanding where insulation, sealing, and handling failures occur is essential to strengthening Thermal Resilience, reducing compliance risk, and building more reliable, climate-ready supply systems.
In practice, many audit programs still focus on three visible checkpoints: data logger records, transport qualification files, and lane validation. Those controls matter, but they do not always reveal whether the shipper wall thickness is sufficient, whether gel packs are conditioned correctly, or whether pallet stacking has created compression damage. A pharmaceutical shipment can remain compliant on paper for 24–48 hours while hidden packaging defects reduce real thermal protection much earlier.
For procurement leaders, QA managers, project engineers, distributors, and operators, packaging weakness is not a minor detail. It influences excursion risk, inventory loss, complaint rates, and audit findings across warehousing, airfreight, passive packaging, and last-mile handover. A stronger audit approach must therefore test the packaging system as an operating asset, not just as a box that accompanies a temperature-controlled product.
Why packaging weak points stay invisible in many cold chain audits
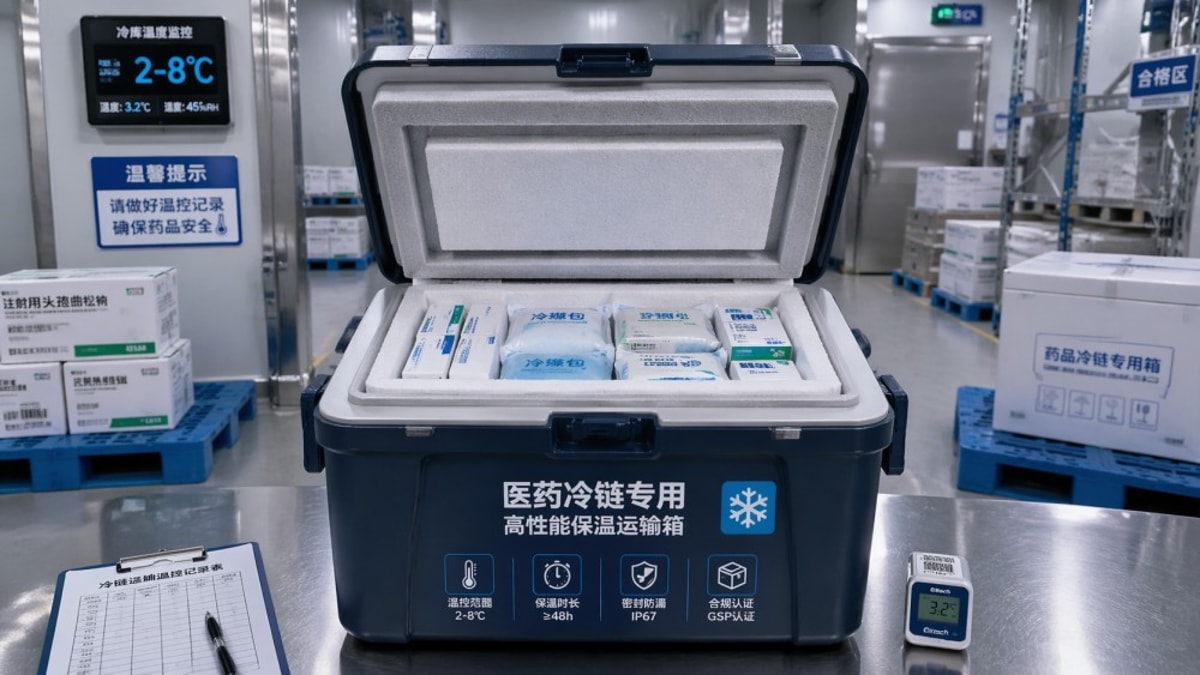
Traditional pharmaceutical cold chain audits are often built around compliance evidence. Auditors review SOPs, training records, route maps, calibration certificates, and deviation logs. These are essential controls, yet they tend to confirm whether a process exists rather than whether the packaging performs under real handling stress. In many operations, the passive shipper is treated as a validated component once approved, then revisited only every 12–24 months.
That gap matters because packaging failure is often cumulative, not immediate. A carton corner crush of 8–15 mm, a small lid gap, or coolant packs loaded in the wrong sequence may not trigger an instant alarm. However, these weak points can reduce hold time by 10%–30% depending on ambient exposure, payload density, and dwell time on the tarmac or loading dock. Standard audits frequently miss this because they do not inspect the physical package after each handoff.
Another issue is sampling bias. Many sites test packaging in ideal conditions: correct pre-conditioning, standard payload weight, and controlled pack-out by trained personnel. Real operations are different. Shift changes, seasonal demand spikes, subcontracted handlers, and mixed-SKU shipping can alter pack-out quality in as little as 1 shift. When audits only examine master procedures, they understate operational variation.
For global enterprises managing vaccines, biologics, APIs, or specialty therapies, this blind spot is amplified by climate volatility. A lane qualified at 25°C mean ambient can behave very differently during a 35°C summer surge, monsoon humidity, or an unexpected 6-hour airport hold. Without packaging-focused inspection, teams may attribute excursions to transport delays alone, when the root cause is insufficient thermal margin in the shipper system.
The audit scope problem
Most audit templates emphasize what is easiest to document and compare across sites. Packaging integrity, however, is more difficult to quantify unless the audit includes physical inspection criteria, handling simulation, and post-transit evaluation. As a result, many enterprises score well on documentation maturity while remaining exposed to avoidable packaging-driven temperature excursions.
Common areas left out of routine audits
- Insulation compression after stacking at 2–4 pallet layers in warehouse staging.
- Seal quality degradation caused by repeated opening, moisture, or tape mismatch.
- Coolant conditioning drift, especially when gel packs or PCM elements vary by more than 2°C from target.
- Payload arrangement errors that create thermal bridges between product and outer wall.
- Returnable packaging wear after 20–50 reuse cycles without structured retirement criteria.
The packaging failure modes that most directly threaten product safety
In pharmaceutical logistics, not all packaging defects have the same impact. The highest-risk weak points usually involve insulation, sealing, coolant placement, and mechanical handling damage. These are the areas most likely to shorten thermal autonomy and create undetected exposure windows during loading, customs dwell, cross-docking, or hospital delivery. Audits that do not review these factors in detail can miss the difference between a 36-hour solution and a real-world 22-hour solution.
Insulation weakness is often underestimated because packaging materials may appear intact. Expanded polystyrene, vacuum insulated panels, polyurethane structures, and corrugated systems all have different failure behaviors. A small puncture in a high-performance panel or moisture ingress into a fiber-based outer structure may not be visible from the outside, but it can significantly alter heat flow. In a lane with 30°C–40°C ambient peaks, a reduced thermal barrier can quickly consume the buffer designed into the shipper.
Sealing is equally critical. Lid fit, tape adhesion, tamper bands, and closure geometry determine air leakage and moisture intrusion. Even a minor closure mismatch can increase internal temperature instability when packages move repeatedly between refrigerated zones and warm handling areas. This is especially relevant for products with narrow ranges such as 2°C–8°C or controlled frozen conditions below -20°C.
Handling damage is the hidden accelerator. A package that passes laboratory qualification may fail in the field after forklift contact, conveyor shock, or corner drop events. For operations shipping hundreds or thousands of boxes per week, damage patterns should be reviewed not as isolated incidents but as predictable stress points. This is where audit teams need packaging engineering input, not just documentation review.
High-risk weak points by packaging function
The table below helps teams connect visible packaging conditions with likely thermal and compliance consequences. It can be used by QA, procurement, warehouse supervisors, and engineering reviewers as a field-level checklist during audits or supplier qualification visits.
A key takeaway is that most failure modes are not independent. A compressed wall can coincide with a poor seal, while incorrect coolant placement can worsen the effect of an already reduced insulation margin. That is why packaging audits should examine the package as a system with at least 4 dimensions: thermal barrier, closure integrity, pack-out accuracy, and handling durability.
Why returnable and reusable systems need extra scrutiny
Reusable pharmaceutical packaging can lower waste and improve consistency, but only if wear is tracked carefully. A returnable shipper used 30 times is not equivalent to one used 5 times. Enterprises should define inspection thresholds such as damaged corners, seal fatigue, latch looseness, or liner deformation, then link those thresholds to retirement or refurbishment actions.
How to redesign audit protocols so packaging performance is measurable
A more effective pharmaceutical cold chain audit treats packaging as a critical control point rather than a passive accessory. This means moving beyond file review and adding measurable field checks. The audit should verify whether the packaging system performs under actual route conditions, operator variation, and handling intensity. In most organizations, this can be implemented through a 5-step enhancement without rebuilding the entire quality framework.
First, define packaging-specific inspection criteria at receiving, pack-out, dispatch, and post-delivery stages. Second, assign thermal risk ratings to each packaging format based on product sensitivity, route duration, and ambient variability. Third, require periodic requalification when lane conditions shift by season or carrier mix. Fourth, document reuse cycle limits for returnable systems. Fifth, trend packaging deviations separately from transport deviations so root causes are not merged into one general exception bucket.
Operationally, these changes are manageable. A site can add 6–10 packaging checkpoints per audit and train reviewers within 2–3 weeks. What matters is consistency. Procurement teams should align packaging specifications with QA expectations, while warehouse operators should use the same acceptance criteria as technical evaluators. Without this alignment, packaging quality becomes subjective and results vary by site.
For global networks, digital evidence also helps. Photos of pack-out configuration, scanned packaging IDs, reuse-cycle logs, and short post-transit inspection forms can provide traceability with limited administrative burden. These records are especially useful when multiple distributors, 3PLs, or regional hubs are involved.
A practical 5-step packaging audit model
- Inspect incoming packaging condition, including wall deformation, seal surfaces, and coolant readiness before use.
- Observe live pack-out for at least 3 shipments per format to confirm orientation, payload spacing, and closure method.
- Check dispatch staging time and ambient exposure, especially when loading docks exceed 15–30 minutes of open exposure.
- Review post-transit packaging condition at destination, not only temperature reports.
- Trend recurring defects monthly and trigger corrective action after defined thresholds, such as 3 similar events in 30 days.
Suggested audit criteria by role
Different stakeholders look for different evidence. The table below helps standardize expectations across QA, operations, procurement, and project management functions.
The strongest audit programs combine these views instead of treating packaging as a narrow warehouse issue. When audit evidence is role-specific but structurally aligned, organizations can move faster from observation to corrective action and supplier improvement.
Procurement, engineering, and operational choices that reduce packaging risk
Better audits are only part of the solution. Enterprises also need packaging selection criteria that reflect route complexity, climate exposure, and handling reality. Procurement teams should avoid choosing packaging only on unit price or nominal hold time. A lower-cost shipper that fails after repeated handling, variable conditioning, or humid storage conditions can create far higher total cost through scrap, investigation time, and emergency reshipment.
A practical sourcing model evaluates at least 4 dimensions: thermal duration under realistic profiles, structural durability, operational ease of pack-out, and supplier support responsiveness. For example, a packaging format validated for 72 hours under controlled testing may still be unsuitable if the site routinely experiences 2-hour outbound staging, mixed courier networks, or multiple open-close events at destination. Technical evaluation should therefore include route mapping and handling observation, not just datasheet comparison.
Engineering teams should also assess compatibility with surrounding cold-chain infrastructure. Packaging performance is shaped by the whole system: refrigerated room stability, dock shelter quality, pallet pattern, AS/RS interfaces, and cryogenic or frozen storage transfer procedures. The packaging that works in one facility may underperform in another if the spatial workflow creates repeated thermal shocks or mechanical stress.
Operator training remains a decisive factor. Even high-performance packaging can underperform if coolant packs are not equilibrated correctly, product is not centered, or closure steps are skipped under time pressure. Sites with frequent temporary labor or distributor-managed pack-out should refresh training every 6–12 months and after any packaging format change.
Selection criteria that deserve more weight
- Thermal autonomy under realistic lane profiles, including summer and winter extremes rather than a single laboratory scenario.
- Tolerance to handling abuse, such as corner impacts, compression, and multiple transfers across 3PL or distributor nodes.
- Pack-out simplicity, because each additional manual step raises error probability during peak-volume periods.
- Inspection and reuse management, especially when returnables circulate across regions with different discipline levels.
- Supplier technical support, including requalification assistance, operator training, and replacement lead times of 7–21 days where needed.
Operational warning signs procurement teams should not ignore
If packaging-related deviations rise during seasonal peaks, if multiple sites improvise with substitute tapes or coolants, or if destination teams report frequent carton deformation, those are not isolated housekeeping issues. They indicate a system mismatch between packaging design and real operation. Early recognition allows procurement and engineering teams to rebalance specification, training, and infrastructure support before excursions escalate.
FAQ: common questions about packaging-focused pharmaceutical cold chain audits
How often should packaging be requalified?
A common interval is every 12–24 months, but that is only a baseline. Requalification should happen sooner if route conditions change, if a new carrier is introduced, if ambient conditions shift materially, or if the packaging design, coolant type, or payload configuration changes. High-risk lanes and reusable systems often justify more frequent review.
What are the most overlooked packaging checks during site audits?
The most overlooked checks are usually post-transit package condition, seal integrity after handling, coolant temperature at pack-out, and visible wall deformation on returnable shippers. Sites also often miss staging-time exposure on loading docks, even though 20–40 minutes of uncontrolled exposure can materially affect thermal margin.
Which teams should own packaging performance?
Ownership should be shared. QA defines acceptance and deviation rules, operations control pack-out execution, procurement manages supplier performance, and engineering or technical evaluation teams verify system suitability. When only one department owns packaging, issues are often discovered late and corrected slowly.
Can packaging issues affect frozen or cryogenic shipments too?
Yes. While the failure mechanisms differ, frozen and cryogenic shipments still depend on containment, insulation continuity, closure integrity, and handling discipline. In these environments, small packaging flaws can cause boil-off changes, dry ice management problems, condensation risk, or unsafe handling conditions if controls are weak.
Pharmaceutical cold chain resilience depends on more than compliant temperatures and complete documents. Packaging weak points in insulation, sealing, coolant management, and handling can quietly erode thermal protection long before an audit exception appears. Organizations that expand audits to include physical packaging evidence, measurable condition checks, reuse controls, and role-based accountability are better positioned to reduce excursions, protect product value, and improve network reliability.
For teams evaluating Cold-Chain Infrastructure, Food Storage Solutions, and Cryogenic Storage strategies, packaging should be reviewed as part of the total thermal system, not as an afterthought. If you need a more rigorous audit framework, packaging evaluation checklist, or infrastructure-aligned cold chain strategy, contact us to discuss a tailored solution and explore more resilient, climate-ready options.
Recommended News




